How Laser Improving Optical Microscopes Sharpness
An optical microscope enables the user to view objects when they are illuminated with light, as opposed to other forms of electromagnetic radiation. Optical microscopes existed long before lasers were created, and they remain an important part of the technology that one uses to view the very small. As with every technology, optical microscopes have certain limitations. In particular, they cannot be used to view objects below a certain size. The quality of the lenses affects the sharpness of the view, but there is another more fundamental restriction: the wavelength of the light used to make the observation.
When features become too small compared to the wavelength of the light used to observe them, those features cannot be resolved. This is called the diffractive limit. The diffractive limit is about one-half wavelength. Violet, which of all the colors has the shortest wavelength, has a wavelength of 400 nanometer (nm). To observe features on an object smaller than 200 nm across, one needs to “look” using much shorter wavelengths. Electron microscopes, for example, are designed to use electrons with extremely short wavelengths. Their resolving power—their ability to distinguish detail—is much better than that of the best optical microscopes, but these instruments are expensive, and the preparation of the specimen is time consuming. Electron microscopes are important tools, but they are poorly suited to certain applications.
An interesting invention, patented in 2004 by a British company called Nanosight, uses a laser, an optical microscope, a computer, and some clever physics to determine the size of particles that are as small as 15 nm across. This is much smaller than the wavelength of any type of (visible) light. To appreciate how small 15 nm is, consider a 15-nm sphere and a billiard ball side by side. (A billiard ball is 2.25 inches [5.71 cm] in diameter.) Now suppose that both objects were expanded the same amount so that the 15-nm sphere became the size of the billiard ball. Under these conditions the billiard ball would expand to a sphere more than 180 miles (300 km) across. Objects 15 nm or even 150 nm across are well beyond the diffractive limit for an optical microscope, but Nanosight’s device, called the Halo LM10, can measure the size of these nanoparticles, which allows the investigator to narrow the possibilities of what the particles in question might be. The system is robust in that specimens do not need a lot of preparation prior to observation, and the system can be used outside the lab. The first step involves placing the sample in water or other fluid and then illuminating the suspension with a laser—in this case, a 20-mil liwatt laser (one milliwatt, abbreviated one mW, equals one-thousandth of a watt).
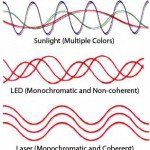
The laser emits red light at a wavelength of 650 nm. At 650 nm, the wavelength of the light used is simply too long to enable the investigator to “see” the particles in the sense that one can observe any distinguishing surface features. Instead, one observes bright spots of light bouncing around on the computer screen. The situation is analogous to observing a swarm of tiny insects under a streetlight. The insects are often so tiny that from a distance one cannot distinguish any detail at all. Their color, wing structure, body structure, and so on are all undetermined. They appear to be undifferentiated dots. Nevertheless, by observing how the swarm moves, one can often correctly deduce the species of insect that one is (not) seeing. Analogously, the laser provides an intense beam of light, the characteristics of which are simple and completely understood, that illuminates the particles. In the light of the laser, one sees bright red dots swarming about one’s field of view with some moving faster than others. In fact, some of the video footage of the laser-illuminated nanoparticles shows them moving about in a way that is strongly reminiscent of a swarm of small insects. The particles are being jostled about the fluid in a phenomenon called Brownian motion. Brownian motion was first observed by Scottish botanist Robert Brown (1773–1858).
Brown noticed that pollen suspended in solution experienced small, rapid and apparently random motions. Although others had noticed the effect before him, Brown was the first to study the phenomenon. During the intervening years, Brownian motion has been studied extensively by mathematicians and physicists.
Today, it is known that the pressure a fluid exerts on a body is the result of numerous random collisions between the body and the molecules (or atoms) of which the fluid is composed. For large bodies, differences in the force or number of collisions averages out over the body’s surface, with the result that pressure seems uniformly distributed over its surface, but the situation is different for small bodies. When a body suspended in a fluid is small enough, there is not sufficient surface area on the body to permit the random collisions that continually occur to “average out.” In such cases, significant, though fleeting, differences in pressure exerted by the molecules along the surface of the particle arise, and as a result the particle undergoes random motion.
Brown and other early scientists searched for a causative agent, something that would enable them to predict the motion of the particles. In one famous experiment, a particle-filled fluid was left sealed in a container for a year, after which time the observations were repeated. The outcome of the experiment was unchanged. Further observations revealed that particle size and fluid temperature and viscosity—a measure of the fluid’s stickiness—are prime factors in determining the motion of the particles. Consequently, if one knows the particle size, one can predict the particle’s motion, and conversely, if one can observe the particle’s motion, one can, in theory, estimate the particle’s size. The latter remark is the observation on which the Halo ML10 is based. If, for example, one suspends the particles in water, where the viscosity and other characteristic properties are known, one can compute what is called the hydrodynamic radius of each illuminated particle by measuring the motion of the particle and the temperature of the water.
Keep in mind that one does not see the particle. Because of its small size, one cannot see the particle, using an optical microscope. One sees only a bright undifferentiated dot. Nevertheless, the motion of the particles yields a measure of particle size. The computed hydrodynamic radius of particles can be combined with observations of the way that the particles present in the fluid scatter light from the laser to produce a fairly precise profile of the sizes of particles present in the fluid. Such profiles are important.
Repeated measurements establish a statistical profile of normal particulate matter in the sampled medium. With this information, researchers are able to determine when new types of particulates are introduced, and all of this can be done in real time. The key is insightful physics, good software, and the right light.