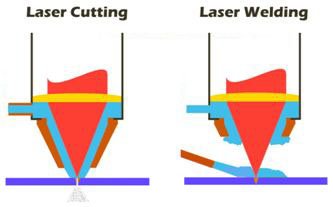
Using Laser for Welding
Welding is another common laser application, but the physics of welding are quite different from the physics of cutting steel and cloth or drilling steel or diamonds. Cutting and drilling generally involve vaporizing small sections of the material in question. By contrast, welding involves melting. The process of welding is in some ways a little more delicate than that of vaporizing: If one applies too much power, the material may vaporize rather than melt; if one applies too little power, the process of conduction causes too much of the energy to dissipate into the surrounding material.
In the latter case, either the energy is conducted away from the target before the metal melts and no melting occurs or, if enough energy is supplied, the melted area becomes too large. Identifying and controlling the many factors that affect a successful laser weld is an important and very active discipline in applied science. Progress has been made—lasers are now an important and widely used tool on automotive assembly lines, for example—but there is still much work to accomplish in understanding and better utilizing this process. The first problem to overcome in making a successful weld involves transferring enough, but not too much, energy to the material.
Keep in mind that metals are often fairly reflective, and electromagnetic radiation that is reflected from the target material is wasted in the sense that it has no effect on the target. However, if it is reflected back at the laser, it may damage the laser itself. In order for the laser to affect the metal, its energy must be absorbed. The absorptivity, which is the percentage of the incident radiation absorbed by the target material, of various common metals depends on a variety of factors, including the finish of the material, its temperature, and the wavelength of the laser beam. In most cases, however, absorptivity is surprisingly low. For example, aluminum at room temperature will initially absorb only about 2 percent of the electromagnetic radiation emitted at a wavelength of 10.6 micrometers (μm), the wavelength characteristic of a CO2 laser, and it will absorb only about 6 percent of incident radiation at a wavelength of 1.06 μm, the wavelength of an Nd:YAG laser, and these are the two most common lasers used in welding. This is one reason that very powerful lasers are used in industrial applications: When the laser beam is first applied to the target, most of the energy has no effect because initially most of the energy is not absorbed. Once the metal melts, however, the absorptivity of the target metal generally increases dramatically. The absorptivity of a CO2 laser beam, for example, jumps from a few percent to 60 or even 80 percent. The melted area begins to deepen, and a pool of liquid metal forms. A great many interesting phenomena occur almost simultaneously at this point and often last for only a fraction of a second.
For example, if the melted area deepens into a V-shape, called a keyhole, the surface of the liquid curves. This concentrates the laser beam toward the center of the region. The energy that is not initially absorbed by the liquid is not generally reflected away from the metal but is instead reflected to the other side of the keyhole, further increasing the absorption of the laser beam. This all happens so fast that a pool of molten metal is formed inside a V-shaped pocket of relatively cool solid metal. The result is that as soon as the laser is no longer directed toward the pool, the heat dissipates into the surrounding region and the pool solidifies. The speed with which a laser weld can be formed has two important implications.
First, lasers can weld seams with amazing rapidity. In a sheet of steel a millimeter thick, for example, a CO2 laser can weld a seam at the rate of 10 meters per minute, much faster than more traditional methods with a concomitant improvement in production rates. Second, because different types of metals conduct heat at very different rates, welding disparate metals together using traditional methods has often proved difficult. Copper, for example, conducts heat at about 10 times the rate of carbon steel, so welding steel onto copper using conventional techniques has proved quite difficult. Heat dissipates much more quickly on one side of the weld than the other. But even in copper, heat conduction happens at a very slow rate compared to the speed with which energy can be introduced via a laser.
As a consequence, welding copper and steel together can be done easily with a laser, because the entire welding process has been completed before much conduction has occurred on either side of the weld. Laser welding is now common in a variety of industrial applications. In some of those applications—welding automobile frames, for example—it has displaced more conventional forms of welding, but in other applications—welding two disparate metals together, for example—it has made processes commonplace that were once difficult or impossible to perform with more conventional implements.


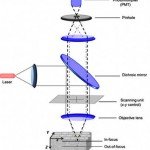 How Laser Improving Optical Microscopes Sharpness
How Laser Improving Optical Microscopes Sharpness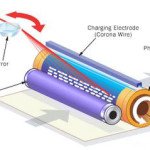 How Laser Printer Works
How Laser Printer Works How to cut metal and diamond using laser
How to cut metal and diamond using laser Laser in Tattoo Removal
Laser in Tattoo Removal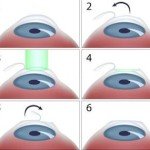 How laser used in eye surgery
How laser used in eye surgery The early history of lasers
The early history of lasers What is Fiber Optics ? How Fiber Optics Works?
What is Fiber Optics ? How Fiber Optics Works?











Hello! I just would like to give a huge thumbs up for the great info you have here on this post. I shall be coming back to your blog for more soon. This answered my problem really, thank you!